WHAT IS ELECTRICITY?
|
|
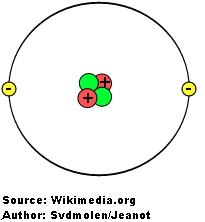
To understand what is electricity, we need to study the atom first. All the matter in the universe is made up of atoms, and atoms are made up of 3 different particles: protons, neutrons and electrons.
|
NEUTRONS
Neutrons are not important to explain electricity, so we will not explain them in this lesson.
Neutrons are not important to explain electricity, so we will not explain them in this lesson.
PROTONS
The number of protons in an atom is very important, because it defines what chemical element the atom represents. For example: an atom of gold has 79 protons and an atom of silver has 47 protons. The number of protons in an atom is called the atomic number. Different atomic numbers, different chemical elements.
The number of protons in an atom is very important, because it defines what chemical element the atom represents. For example: an atom of gold has 79 protons and an atom of silver has 47 protons. The number of protons in an atom is called the atomic number. Different atomic numbers, different chemical elements.
ELECTRONS
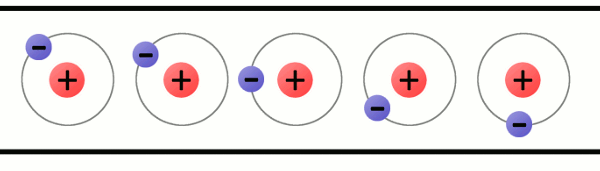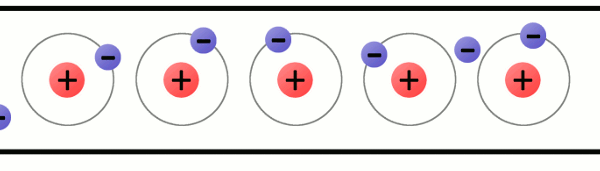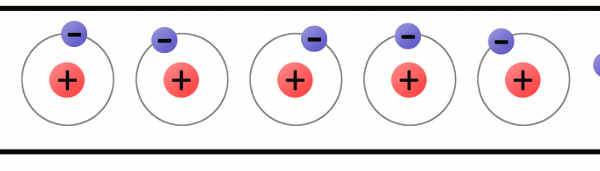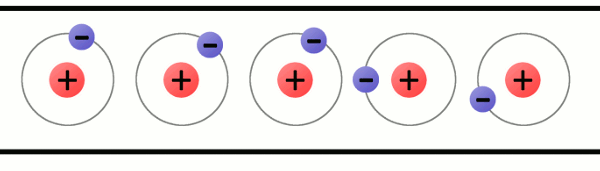
Electrons are very special because they are not permanent attached to atoms. They can move from one atom to another. How can an atom lose an electron? When materials rub together. When an atom has more protons than electrons, we say that the atom has a positive charge; while when an atom has more electrons than protons, we say that it has a negative charge.
Electrons are very special because they are not permanent attached to atoms. They can move from one atom to another. How can an atom lose an electron? When materials rub together. When an atom has more protons than electrons, we say that the atom has a positive charge; while when an atom has more electrons than protons, we say that it has a negative charge.
The problem is that atoms like to be stable, that is, they like to have the same amount of protons and electrons.
So, when an atom is unbalanced, when it loses an electron, it will look for an electron to be balanced again.
How can an atom get a new electron? It will steal an electron from another atom. Then, another atom will be unbalanced and that process will start again.
That is what we call electricity: electricity is the flow of moving electrons thanks to unbalanced atoms.
So, when an atom is unbalanced, when it loses an electron, it will look for an electron to be balanced again.
How can an atom get a new electron? It will steal an electron from another atom. Then, another atom will be unbalanced and that process will start again.
That is what we call electricity: electricity is the flow of moving electrons thanks to unbalanced atoms.
That flow of moving electrons, that is, electricity, can be conducted better through some materials than through another.
Some materials can lose electrons very easily. We call these materials electrical conductors. Metals, water and people are good electrical conductors.
On the other hand, some materials hold their electrons very tightly, so it is difficult for them to lose electrons. We call these materials electrical insulators. Glass, plastic, porcelain and rubber are good electrical insulators.
Some materials can lose electrons very easily. We call these materials electrical conductors. Metals, water and people are good electrical conductors.
On the other hand, some materials hold their electrons very tightly, so it is difficult for them to lose electrons. We call these materials electrical insulators. Glass, plastic, porcelain and rubber are good electrical insulators.
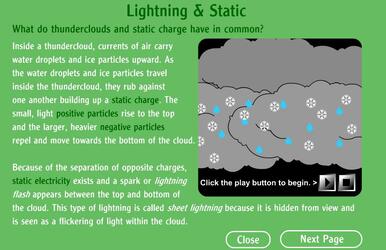
On the following animation you will check how thunders are created. Click on the picture.
What about watching some videos to get more information about electricity?
On the first video you will review what is electricity.
On the first video you will review what is electricity.
On the second and the third videos you will learn how to build a simple electric motor.
On the fourth video you will learn how to build an electric train using batteries and copper wire.