HOW DOES HEAT CHANGE OBJECTS
|
|
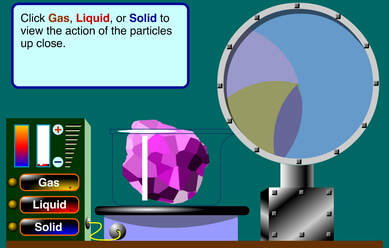
Once we have learned what is heat and how does it travel, it's time to learn how heat affect the objects. But first, we should review how are molecules in a solid, a liquid and a gas organized. Click on the picture to start the animation.
|
When we add or take away thermal energy to an object, it can happen 3 things: that object can expand or contract, the object can suffer a change of state or the object can suffer a chemical change.
EXPANSION AND CONTRACTION
If we add thermal energy to an object, its molecules will begin to vibrate more vigorously, so they will need a little more space. As a result, expansion takes place. Liquids expand more than solids, and gases expand more than liquids. On the other hand, if we take away energy from an object, its molecules will begin to move less vigorously, so they will need less space. As a result, contraction takes place.
EXPANSION AND CONTRACTION
If we add thermal energy to an object, its molecules will begin to vibrate more vigorously, so they will need a little more space. As a result, expansion takes place. Liquids expand more than solids, and gases expand more than liquids. On the other hand, if we take away energy from an object, its molecules will begin to move less vigorously, so they will need less space. As a result, contraction takes place.
CHANGE OF STATE
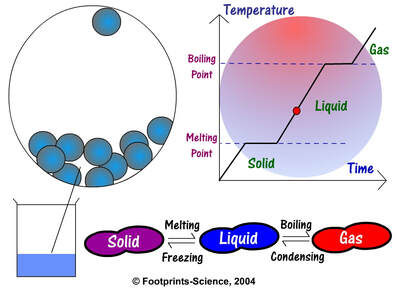
The most important ones are: melting, freezing, evaporation and condensation.
The most important ones are: melting, freezing, evaporation and condensation.
- Melting: if we add enough thermal energy to a solid, it will change into a liquid. How is that possible? Heat makes the molecules in the solid move faster, so molecules move away from each other and this action changes the physical appearance of the object.
- Freezing: if we take away enough energy from a liquid, it will change into a solid. How? Less thermal energy means that the molecules in the liquid will move slower, so they will settle down. Freezing and melting are opposite processes.
- Evaporation: if we add enough energy to a liquid, it will change into a gas. This process is very similar to melting: heat makes the molecules in the liquid move faster, so they move away from each other and the physical appearance of the object changes.
- Condensation: if we take away enough energy from a gas, it will change into a liquid. This process is very similar to freezing: less thermal energy means that the molecules in the gas will move slower, so they will settle down. Evaporation and condensation are opposite processes.
CHEMICAL CHANGE
When a substance suffers a chemical change, a new substance is formed. Thermal energy can cause chemical changes, for example, when cooking: if we cook an egg, it is transformed from a thick liquid into a solid. Chemical changes can't be reversed: we can't change back the solid egg into a thick liquid again.
When a substance suffers a chemical change, a new substance is formed. Thermal energy can cause chemical changes, for example, when cooking: if we cook an egg, it is transformed from a thick liquid into a solid. Chemical changes can't be reversed: we can't change back the solid egg into a thick liquid again.
Let's ease the states of matter explanation a little more with the following animation. Click on the picture.
What about watching a video about expansion and contraction?
Do you like games? So go to the games section to check what have you learnt in this lesson.